Wellness
January 6, 2026
Healthy Reset After the Holidays
As the decorations come down and schedules return to normal, now is the perfect time to reset, not punish yourself. Instead of jumping straight into strict resolutions, focus on restoring healthy habits that support your overall well-being for the long term.

August 1, 2025
Finding Hope for Opioid Addiction at CRMC
CRMC’s Hope Clinic offers Suboxone therapy in Crosby and Longville to support those battling opioid addiction. “Addiction affects so many families, and people deserve recovery care close to home,” said Dr. Lacey Running Hawk. Trained CRMC providers are committed to guiding patients toward a safer, healthier future.

June 30, 2025
Summer Safety Tips to Avoid ER Visits
Stay safe this summer with tips on drowning prevention, running, biking, and heat exhaustion. Avoid injuries and make the most of the season.

The Healing Touch: Osteopathic Manipulative Medicine
Discover how Dr. Courtenay Deane, D.O., uses gentle touch to ease pain, restore motion, and support healing with osteopathic care at CRMC.

May 1, 2025
Don’t let Allergies Cloud your Spring
CRMC Pediatrician Wendi Johnson, MD, FAAP, recommends taking a proactive approach to allergy season to stay ahead of those sniffles and sneezes.
April 2, 2025
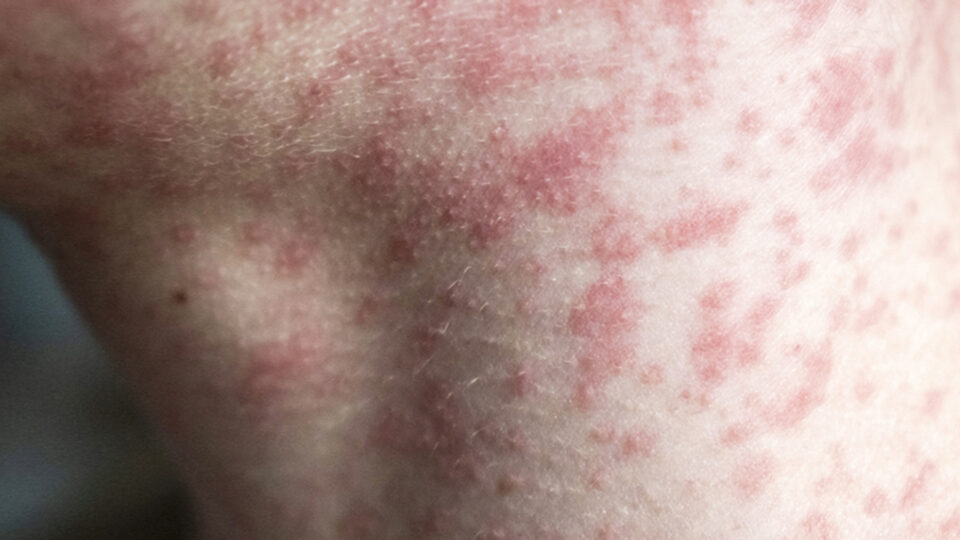
Stay Informed, Stay Protected: What You Need to Know About Measles
As Measles cases continue to pop up across the nation, you may be left feeling like you have more questions than answers. CRMC Pediatrician Wendi Johnson, MD, FAAP, shares some of the must-know facts surrounding the recent Measles outbreak.

February 23, 2025
Cancer Prevention: Human Papillomavirus (HPV) and why Vaccination Matters
HPV is a common virus that can lead to serious health complications in both men and women causing cervical and throat cancer, as well as other cancers. HPV is responsible for 90% of anal and cervical cancers and 70% of oropharyngeal cancers.

February 3, 2025
Don’t Let Seasonal Depression Go Unnoticed

October 30, 2024
Is it the cold, flu, or RSV?
Cold and flu season often packs a punch with symptoms like fever, sore throat, and congestion. No one wants to feel under the weather, especially with holiday gatherings and seasonal activities keeping schedules jam-packed. So, if you’re feeling crummy, what’s to blame? Is it the common cold, or could it be something more?

Coping with Holiday Stress
The hustle and bustle of the holiday season, often filled with joy and anticipation, can quickly turn dismal and dull. Before you know it, you’re left frazzled. Behavioral Health Psychologist Kristin Furan, PsyD, has some tips to help you cope with holiday stress.

